|
An interactive periodic table may be found here. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.\): The modern version of the periodic table. Figure 2 2: Within each period, the trend in atomic radius decreases as Z increases for example, from K to Kr. The concept of electronic configuration has replaced the older concept of valency and valence electrons. Group 2 elements, the alkaline earth metals, have a valency of +2 as they tend to lose two. The electronic configuration of each element is decided by the Aufbau principle which states that the electrons fill orbitals in order of increasing energy levels. So the outermost shell contains 5 valence electrons. The 1s shell contains 2 electrons, the 2s shell contains 2 electrons and the 2p shell contains 6 electrons. The atomic number of a Phosphorous atom is 15. This means they readily lose one electron to achieve a stable octet configuration. Phosphorus Valence Electrons Dot Diagram. For example, elements in Group 1 of the periodic table, known as alkali metals, have a valency of +1. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). The stronger pull (higher effective nuclear charge) experienced by electrons on the right side of the periodic table draws them closer to the nucleus, making the covalent radii smaller. Valency varies for different elements across the periodic table. 
Free Gift for you: Interactive Periodic Table The electron configuration of a oxygen atom is. While core electrons are not involved in bonding, they influence the chemical reactivity of an atom. Atoms have a tendency to have eight electrons in their valence shell. Valence electrons determine the reactivity of an atom. If your periodic table doesn't already have each column numbered, give each a number starting with 1 for the far left end and 18 for the far right end. Generally, valence electrons can participate in the formation of chemical bonding, but core electrons cannot. Valence electrons are the outer-shell electrons of an atom. Generally, on a periodic table, all of the elements in a single vertical column will have the same number of valence electrons. Note 2: If you want a periodic table with valence electrons labeled on it, then visit this article: Periodic table with Valence electrons (labeled on it)(From this article, you can also download the HD image of periodic table labeled with valence electrons, so visit right now). Label each column on the periodic table of elements from 1 to 18. Note 1: If you want the valence electrons of all the 118 elements, then visit this article: Valence electrons chart for ALL ELEMENTS(Where I have shown the valence electrons using images). Valence electrons: The electrons present in the outermost orbit of an atom are called valence electrons. 
The reason why this electron configuration seems more complex is that the f-block, the Lanthanide series, is involved. 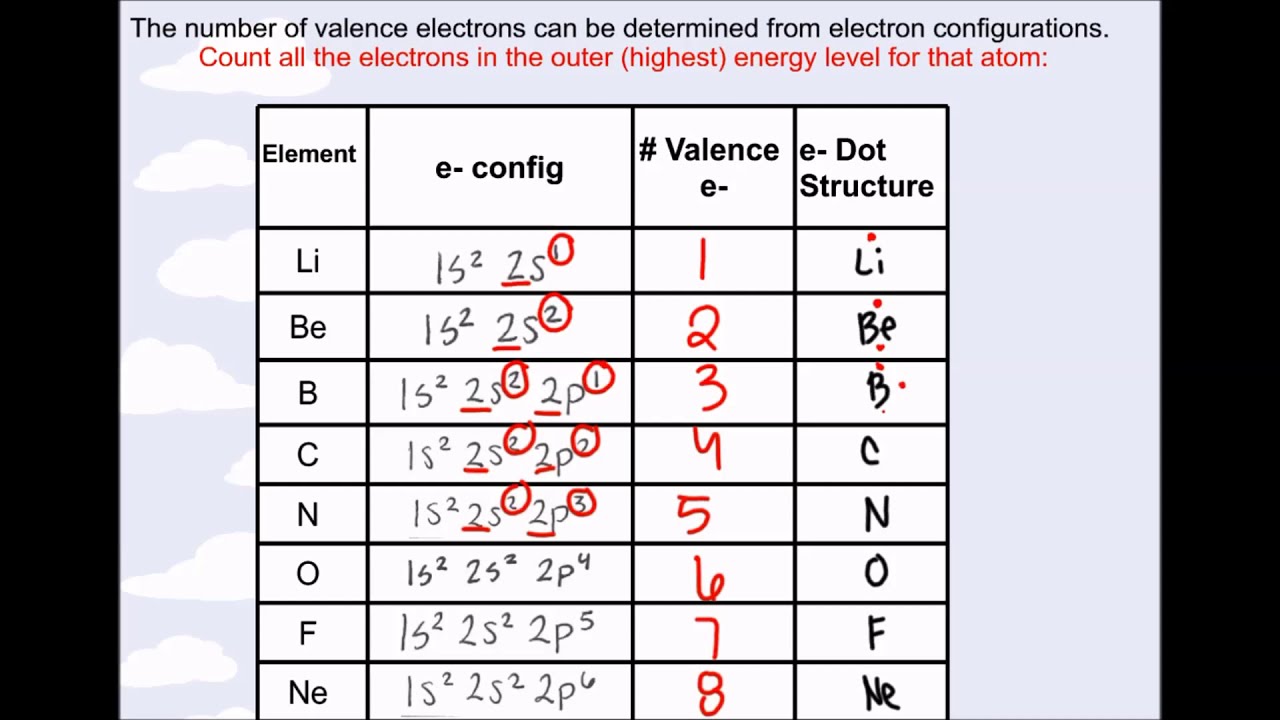
Atomic Number Element Valence electrons 1 Hydrogen (H) 1 2 Helium (He) 2 3 Lithium (Li) 1 4 Beryllium (Be) 2 5 Boron (B) 3 6 Carbon (C) 4 7 Nitrogen (N) 5 8 Oxygen (O) 6 9 Fluorine (F) 7 10 Neon (Ne) 8 11 Sodium (Na) 1 12 Magnesium (Mg) 2 13 Aluminum (Al) 3 14 Silicon (Si) 4 15 Phosphorus (P) 5 16 Sulfur (S) 6 17 Chlorine (Cl) 7 18 Argon (Ar) 8 19 Potassium (K) 1 20 Calcium (Ca) 2 21 Scandium (Sc) 2 22 Titanium (Ti) 2 23 Vanadium (V) 2 24 Chromium (Cr) 1 25 Manganese (Mn) 2 26 Iron (Fe) 2 27 Cobalt (Co) 2 28 Nickel (Ni) 2 29 Copper (Cu) 1 30 Zinc (Zn) 2 The periodic table gives the following electron configuration: 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 10 5p 6 6s 2 4f 14 5d 10 6p 3. Valence electrons of first 30 elements of the Periodic table are mentioned below.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
